An adverse reaction is defined in the regulations as a microchip that causes any unnecessary pain or suffering or any pathology that is or seems to be caused by the implantation of a microchip, or a microchip that has migrated from the implantation site or failed (failure to transmit the number encoded in the microchip when scanned by an appropriate transceiver). Although the regulation only applies to dogs (and in England, cats) it would be good practice to report adverse reactions to microchips in other species as well. Similarly, although the Northern Ireland legislation does not make a specific requirement to report adverse reactions, the BSAVA recommends these should be reported.
The regulations in England, Scotland and Wales make it a legal requirement to report adverse reactions/events relating to microchips and microchip failure in dogs, and in England this also applies to cats from 10th June 2024. Where there is evidence of any adverse event in relation to a microchip this must be reported to Defra using the microchipping adverse event form. Although this requirement applies to everyone, it is likely that those in veterinary practices may be the first to become aware of these problems, because they regularly scan microchips. The RCVS has now included guidance that veterinary surgeons and veterinary nurses should report any adverse reaction to a microchip or the failure of a microchip in the supporting guidance to the Code of Professional Conduct.
The Scottish Regulations require reports to be made within 21 days beginning with the day the adverse reaction, migration or failure is identified.
If you are in contact with the microchip provider, they may offer to report the adverse reaction to Defra on your behalf; in order to facilitate this make sure you have all the details to hand to pass on to them. Where the report of an adverse event is not made directly by the veterinary practice we recommend that you record arrangements for reporting, for future reference.
It is important to report all adverse reactions/events in order to monitor any problems with microchips or implantation standards so that steps can be taken to resolve any problems.
- Further details of the scheme can be found on the Gov.uk website
- Reports should be submitted by emailing the microchipping adverse event form to microchipping@defra.gov.uk
- Queries can be addressed to Defra at: microchipping@defra.gov.uk.
Types of adverse event
Please note that while the legislation refers to ‘adverse reactions’ the Defra reporting scheme refers to ‘adverse events’. In this document we have treated these terms as synonymous.
Implantation reactions
These usually occur relatively soon after implantation and are commonly linked to the implantation procedure e.g. haematomas (bleeding under the skin) or infection (an abscess near the site or an infection spreading systemically, making the animal ill).
At the time of implanting the microchip, it is not unusual for there to be an initial pain response, and sometimes a little bleeding at the implantation site. This should stop within a minute or two and would not be considered as an adverse event.
On rare occasions, an animal may react to an implanted microchip, as with any foreign material introduced into the body, with an inflammatory response. These along with any evidence of infection or other pathology should be reported as adverse events.
There are a very small number of reports of paralysis resulting from implantation into the spinal cord. Although these relate to problems with implantation technique rather than a problem with the microchip, these should also be reported. There have also been a very small number of cancers reported around the site of microchip implantation. Although these may or may not be related to the implantation of the microchip (c.f. feline injection site sarcomas) these should still be reported so that Defra is able to collect accurate data on the incidence of these types of reaction.
Microchip Migration
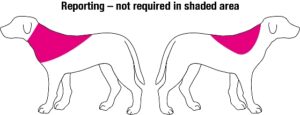
Migration from the site of implantation is classed as an adverse event, and there is some logic in this in that it enables problems with certain brands or batches of microchips, or with the technique of certain implanters, to be detected. However, as there is no requirement to record the site of microchip implantation on the animal’s record or microchip database, actual migration – as opposed to incorrect placement at initial implantation – is difficult to ascertain. For this reason we recommend that unless the veterinary surgeon has more detailed evidence on implantation site, for example from their own records or through entry in a Pet Passport, microchips located within the areas indicated in red on the diagram below, which covers the recommended UK and European sites for implantation in both dogs and cats (see Implantation section for recommended sites) do not need to be reported as having migrated, allowing for minor errors of positioning at implantation.
Microchip Failure
Before assuming a microchip has stopped working it is good practice to ensure that the scanner is fully powered and working correctly and is compatible with the microchip in the animal (some microchips used in other countries will not work with all scanners available in the UK). The whole animal should be scanned and re-scanned in case the microchip has moved from the implantation site or has been implanted in a different part of the dog. If in doubt, try using a different scanner. If the microchip still cannot be read an attempt should be made to palpate the microchip under the skin or, if the owner wishes to confirm the presence of an unresponsive microchip, diagnostic imaging such as radiography or ultrasound can be used. It is worth noting that the most common reason for failing to detect an implanted microchip is that it migrated out of the animal shortly after implantation.
The veterinary surgeon is only expected to report microchip failure where they have some evidence that a microchip had been present, for example through a registration document or patient record that provides details of a microchip number. As the microchip number is essential for reporting to Defra the owner should be strongly encouraged to locate this information.
If an owner presents an animal without a working microchip and with no evidence of a microchip having been present, the veterinary surgeon can offer diagnostic imaging to ascertain if a microchip is present. Please note that members of the Microchip Trade Association are only likely to consider payment for imaging if a non-functioning chip is detected (see https://microchiptradeassociation.org/page8/page5/ ).
Microchip failure – practical considerations
With the introduction of compulsory microchipping, an animal with a failed microchip will need to have a new microchip implanted unless it qualifies for exemption (as detailed in the Requirements and Exemptions section). Where a second microchip is implanted it is recommended that the veterinary surgeon records details of the reason for re-implantation and where appropriate provides a written declaration to the owner with information about both the old and new microchip.
Failed microchips and travel documentation
Given the potential implications if a microchip fails on entry to the UK – such as the cost to the owner of resulting time in quarantine – veterinary surgeons should encourage their clients to have their pet’s microchip checked before travel. BSAVA strongly recommends that owners and vets check the Defra website for up-to-date requirements for travel: https://www.gov.uk/bring-pet-to-great-britain.
Defra provides the following advice on unreadable microchips:
- If the original microchip cannot be read, the pet must be re-implanted;
- If the pet has been previously prepared for travel, then it must be re-prepared in accordance with pet travel rules and new travel documentation (such as an Animal Health Certificate) issued;
- The issuing veterinary surgeon should note in the ‘Marking of animals’ section of the Animal Health Certificate that there is another microchip present (and the number of that microchip) that cannot be scanned and read, in case the fault is intermittent and the chip is picked up at the port when the pet is checked for compliance for entry to the UK.
Intermittently working microchips and travel documentation
If the microchip has not failed completely, Defra recommends that a new microchip is inserted in case the existing microchip fails at some point in the future. The following procedure must be carried out in order to maintain a pet’s compliance:
- The original microchip, implanted prior to the initial rabies vaccination, must be scanned and a reading made. If a reading is not possible it will be necessary to complete the procedures for a failed microchip;
- Having gained a reading, a new microchip may then be inserted. In cases where a second microchip is inserted, in the ‘Marking of animals’ section of the Animal Health Certificate the Official Veterinarian should write: “On (date), I scanned and read microchip number (original). Upon gaining a reading, I inserted microchip number (new) on the same day, in accordance with Defra requirements” (or similar wording).
This statement does not need to be exactly as shown, but the dates of microchip readings and the numbers of the microchips must be stated, and this statement should be signed and stamped by the OV. Provided this procedure is carried out as stated above, the pet will remain fully compliant with UK entry requirements under pet movement rules.
Removing microchips
It is not necessary to remove a failed microchip, unless it is associated with some pathology. The RCVS provides the following guidance on the removal of microchips:
- 29.25 Because of the importance attached to the accurate identification of animals and the potential for fraud, a microchip must only be removed where this can be clinically justified. This justification should be documented and where required another microchip or alternative method of identification used;
- 29.26 Removal of a microchip in any other circumstances would be an unnecessary mutilation. While the insertion of a second microchip may be problematic, this in itself does not justify removal of a microchip and an audit trail must be maintained.
