Preoperative microbial load considerations
2 November 2022
Rebecca Verhoven BSc (Hons) RVN and lecturer Kat Hart report on their PetSavers-funded research undertaken while Rebecca was a student vet nurse – Cotton wool versus non-woven gauze swabs: comparatively, which is more efficacious for minimizing the preoperative microbial load on the ovariohysterectomy surgical site in bitches?
In 1867, armed with Louis Pasteur’s ‘germ theory’, British surgeon Joseph Lister pioneered the principles of antisepsis: the eradication of transient skin flora and the inhibition of resident microorganisms, by the use of carbolic acid (phenol) as an antiseptic to prevent surgical sepsis.1 As a result, the rate of surgical site infection (SSI) declined rapidly, catalysing wider acceptance and adoption of antiseptic practices over time.2 Nevertheless, SSI will remain an ever-persistent threat in veterinary and human surgery, due to the inevitable compromise of the host’s physical defences, creating a portal for infective microorganisms to invade and multiply.3
SSIs have been reported as a complication in 0.8% to 18.1% of veterinary surgeries, with rates varying considerably according to wound classification.4,5,6,7 Studies show commensal bacteria to be the leading cause of SSI, with Staphylococcus pseudintermedius strains most frequently isolated from infection sites in dogs.8 The consequences of these infections may include prolonged hospitalization, reconstructive surgery, extended wound management, antibiotic resistance, increased health care costs, emotional burden on the owner and practitioner, and patient mortality.7,9 Hence,some researchers exaggerate the importance of implementing an effectual and well-established pre-surgical skin antisepsis protocol to prevent opportunistic pathogens from causing infection.3,10,11
The antiseptic agent and application technique have been recognized as the two key components of surgical skin preparation. Both elements have been researched and debated extensively within the human and veterinary sector, enabling evidence-based recommendations to be made;12 however, there has been little investigation into the material utilized to apply the antiseptic solution. Although a number of opposing opinions have been expressed on the efficacy of cotton wool and non-woven gauze swabs to perform the preparatory surgical scrub, in veterinary research there is no scientific evidence to support such claims. Furthermore, the association between the incidence of wound interference behaviours from patients postoperatively and the application material chosen to clean the skin pre-surgically has not been investigated.
Study aims
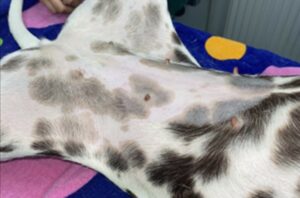
The objectives of this study were to investigate the clinical efficacy of cotton wool versus non-woven gauze swabs for reducing microbial contamination of the ovariohysterectomy surgical site in bitches pre-operatively (Figure 1), measured by counting the colony forming units (CFU/ml) on swabs collected pre- and post-antisepsis. Additionally, it aimed to explore the effect of the material utilized for skin antisepsis on the prevalence of post-operative wound interference behaviours.
Study findings
All swab samples collected post-antisepsis presented without positive microbial growth (0 CFU/ml), confirming no statistically significant difference between the two materials in reducing the number of microbes on the skin. The Fisher’s exact test identified no significant difference for post-operative licking of the surgical site between the standard treatment and experimental group (p = 0.24). Therefore, it was determined that cotton wool and non-woven gauze swabs were equally effective at reducing the number of microorganisms on the skin, and subsequently minimizing the risk of SSI. Additionally, neither cotton wool nor non-woven gauze swabs were an observed risk factor for post-operative patient interference.
Importance of the findings
This research will add value to the current evidence base within veterinary science to provide professionals with a long-awaited answer regarding appropriate skin cleansing materials. This may encourage practices to update their current standard operating procedures in this area of clinical practice to promote consistency and evidence-based patient care. Furthermore, if practices were to be consistent in the material used to clean the pre-operative skin, this may reduce the likelihood of this aspect needing to be considered as a risk factor for SSI when staff conduct clinical audits in this area. We hope our research is also shared by veterinary teaching staff to provide the next generation of veterinary nurses and veterinary surgeons with evidence-based answers.
Applying for a BSAVA PetSavers grant and carrying out the study
The COVID-19 pandemic was the ultimate challenge I faced during my study. In line with Government guidance during the third national lockdown, veterinary practices were advised by the RCVS to provide urgent care and emergency treatment only. Therefore, recruitment of a sufficient number of cases for my study was particularly challenging in a delimited time-frame. Despite the difficulties, my first experience with research has taught me a number of skills including report writing, data collection, and
critical thinking, which I look forward to developing in the future.
On submission of my application to PetSavers, approval was not granted immediately; however, PetSavers was quick to communicate constructive feedback and offer additional guidance on suggested revisions. Support was readily available, and I was fortunate enough to receive a Zoom call from a member of the team to discuss the committees’ comments to move forward with my project. I am so thankful to PetSavers for seeing value in my proposal and encouraging me to resubmit a successful application.
The advice I would offer to any student thinking of applying for a BSAVA PetSavers research grant would be to get organized and prepare for submission ahead of time to avoid unnecessary stress, do not hesitate to ask questions if you are unsure, and to reach out to more experienced individuals in your chosen field of study for advice. Most importantly, believe in yourself and your research and the PetSavers team will do the same.
References
1.Dockery G (2012) Surgical principles. In: Lower Extremity Soft Tissue and Cutaneous Plastic Surgery, 2nd edition, eds. G Dockery and ME Crawford, pp. 23–27. Philadelphia: Saunders DOI: 10.1016/B978-0-7020-3136-6.00004-7 https://doi.org/10.1016/B978-0-7020-3136-6.00004-7
2.Hemani ML and Lepor H (2009) Skin preparation for the prevention of surgical site infection: which agent is best? Reviews in Urology 11 (4), 190–195. Google Scholar
3.Melekwe GO, Uwagie-Ero E, Zoaka HA and Odigie EA (2018) Comparative clinical effectiveness of preoperative skin antiseptic preparations of chlorhexidine gluconate and povidone iodine for preventing surgical site infections in dogs. International journal of Veterinary Science and Medicine 6 (1), 113–116; doi: 10.1016/j.ijvsm.2018.03.005 https://doi.org/10.1016/j.ijvsm.2018.03.005
4.Vasseur PB, Levy J, Dowd E and Eliot J (1988) Surgical wound infection rates in dogs and cats: data from a teaching hospital. Veterinary Surgery 17 (2), 60–64; doi: 10.1111/j.1532-950X.1988.tb00278.x https://doi.org/10.1111/j.1532-950X.1988.tb00278.x
5.Nicholson M, Beal M, Shofer F and Brown DC (2002) Epidemiologic Evaluation of Postoperative Wound Infection in Clean-Contaminated Wounds: A Retrospective Study of 239 Dogs and Cats. Veterinary Surgery 31 (6), 577–581; doi: 10.1053/jvet.2002.34661 https://doi.org/10.1053/jvet.2002.34661
6.Eugster S, Schawalder P, Gaschen F and Boerlin P (2004) A Prospective Study of Postoperative Surgical Site Infections in Dogs and Cats. Veterinary Surgery 33 (5), 542–550; doi: 10.1111/j.1532-950X.2004.04076.x https://doi.org/10.1111/j.1532-950X.2004.04076.x
7.Turk R, Singh A and Weese JS (2015) Prospective Surgical Site Infection Surveillance in Dogs. Veterinary Surgery 44 (1), 2–8; doi: 10.1111/j.1532-950X.2014.12267.x https://doi.org/10.1111/j.1532-950X.2014.12267.x
8.Sasaki T, Kikuchi K, Tanaka Y, Takahashi N, Kamata S and Hiramatsu K (2007) Reclassification of Phenotypically Identified Staphylococcus intermedius Strains. Journal of Clinical Microbiology 45 (9), 2770–2778 DOI: 10.1128/JCM.00360-07 https://doi.org/10.1128/JCM.00360-07
9.Nelson LL (2011) Surgical Site Infections in Small Animal Surgery. The Veterinary Clinics of North America: Small Animal Practice 41 (5), 1041–1056; doi: 10.1016/j.cvsm.2011.05.010 https://doi.org/10.1016/j.cvsm.2011.05.010
10.Belo L, Serrano I, Cunha E, Carneiro C, Tavares L, Carreira L and Oliveira M (2018) Skin asepsis protocols as a preventive measure of surgical site infections in dogs: chlorhexidine–alcohol versus povidone–iodine. BMC Veterinary Research 14 (1), 95; doi: 10.1186/s12917-018-1368-5 https://doi.org/10.1186/s12917-018-1368-5
11.Reynolds H and Nichols A (2019) Which skin preparation technique is most effective to minimise bacterial contamination? The Veterinary Nurse 10 (3), 162–166; doi: 10.12968/vetn.2019.10.3.162 https://doi.org/10.12968/vetn.2019.10.3.162
12.Tanner J (2012) Methods of skin antisepsis for preventing SSIs. Nursing Times 108 (37), 20–22. Google Scholar
